Medical Device EMC Testing
Stancer Testing-Lab offers testing and certification services for a wide-range of medical devices. Our services are mainly focused on EMC/RF compliance testing and certification of medical devices in accordance with applicable international standards.

- Industries
- Medical
Electromagnetic compatibility (EMC) testing is a critical component of medical device design, validation, and regulatory approval. Proper electromagnetic compatibility ensures that medical equipment performs safely and reliably in real world electromagnetic environments. Devices must resist interference from surrounding systems while limiting their own electromagnetic emissions. Effective EMI testing and RF testing directly impact patient safety, device reliability, and regulatory clearance.
In the United States, the FDA outlines expectations for EMC testing and radio frequency testing within medical device submissions. These expectations integrate international standards and risk based validation principles. Manufacturers must demonstrate compliance through structured test data aligned with recognized standards such as IEC 60601-1-2, the global benchmark for medical device EMC performance.
Regulatory Framework for Medical Device EMC Testing
Medical devices are regulated under the Federal Food, Drug, and Cosmetic Act. Products containing electronic circuitry including infusion pumps, patient monitors, imaging systems, and wearable therapeutic devices must demonstrate both:
- Controlled electromagnetic emissions through EMI testing
- Immunity to external disturbances through RF testing and transient testing
Recognized international standards include IEC 60601-1-2 for EMC requirements and testing, the IEC 61000-4 series for electromagnetic test methods, and ISO 14971 for medical device risk management.
IEC 60601-1-2 Core Standard for Medical EMC Compliance
IEC 60601-1-2 defines emission limits and immunity criteria to ensure essential performance is maintained under electromagnetic stress conditions.
Emission Limits
Limits for conducted and radiated emissions prevent interference with nearby equipment, wireless systems, and communication networks.
Immunity Requirements
- Electrostatic discharge according to IEC 61000-4-2
- Radiated RF immunity according to IEC 61000-4-3
- Electrical fast transients according to IEC 61000-4-4
- Surge immunity according to IEC 61000-4-5
- Conducted RF immunity according to IEC 61000-4-6
- Voltage dips and interruptions according to IEC 61000-4-11
Integrating EMC Testing into Risk Management
Under ISO 14971, manufacturers must identify electromagnetic hazards, assess their severity, and implement mitigation strategies. EMC disturbances such as RF exposure, surge events, switching noise, and electrostatic discharge can compromise device functionality or affect clinical performance. Test levels should be selected based on intended use environment, proximity to wireless infrastructure, and safety classification of the device.
Global Regulatory Considerations
Manufacturers seeking access to European markets must consider compliance with the EMC Directive 2014/30/EU and the RED Directive 2014/53/EU for devices incorporating intentional radio transmitters. Wireless medical devices may require evaluation under both IEC 60601-1-2 and RED requirements to support CE marking.
Strategic Importance of EMC and RF Testing
EMC testing, EMI testing, and radio frequency testing are fundamental to regulatory approval and patient safety. Structured laboratory evaluation ensures medical devices operate safely within complex healthcare electromagnetic environments while meeting global compliance expectations.
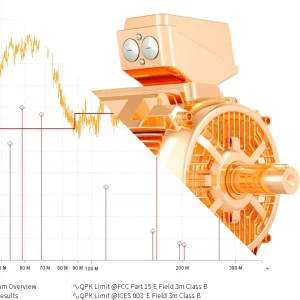
Broadband Radiated Emissions Caused by a Brushed DC Motor

FDA EMC Guidance for Medical Devices

Automotive EMC Emission and Compliance Testing

Regulatory Compliance for IoT

Key Reasons for EMC test failure

Why Wireless Products Fail Regulatory Compliance in the Real World

Vehicle Electromagnetic Compatibility (EMC) Regulatory Compliance and Practical Implementation

Understanding EU Harmonized Standards for EMC and Product Compliance
Have Questions? Let’s talk
Get in touch, and we’ll be happy to assist you with your questions or needs.







